
ISO 20417:2026 / Medical devices manufacturer information
ID 25792 | 19.03.2026 / Preview attached
ISO 20417:2026
Medical devices - Information to be supplied by the manufacturer
Published (Edition 2, 2026)
Publication date: March 2026
This second edition cancels and replaces the first edition (ISO 20417:2021), which has been technically revised.
__________
This document provides general requirements for the identification and labelling of a medical device or accessory that appears on the packaging, is marked on the medical device or accessory and is contained in the accompanying information. The aim of this document is to serve as a central source of these common, generally applicable requirements, allowing each specific product standard or group standard to focus more concisely on the unique requirements for a specific medical device or group of medical devices.
The requirements of a medical device product standard or a group standard can make use of these general requirements to simplify those documents. Additional specific product information requirements can be set out in specific product standards or group standards. Unless specified otherwise within a product standard or a group standard, the general requirements of this document apply.
This document has been prepared in consideration of:
- the application of Essential Principles of Safety and Performance of Medical Devices and IVD Medical Devices, IMDRF/GRRP WG/N47:2024 on the information supplied by the manufacturer of a medical device (see Annex D);
- the application of Labelling Principles for Medical Devices and IVD Medical Devices, IMDRF/GRRP WG/N52:2024 on the information supplied by the manufacturer of a medical device (see Annex D);
...
This second edition cancels and replaces the first edition (ISO 20417:2021), which has been technically revised.
The main changes are as follows:
- update of the normative references;
- deletion of the former informative Annexes D, F, G and H;
- addition of the term ‘applicable policy’;
- deletion of item b) in Clause 4, and of item d) 1) in 6.1.2.
...
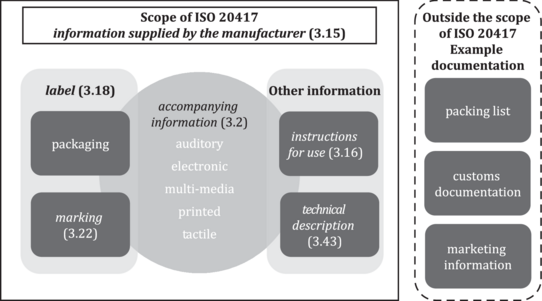
Figure 1 - Relationship of terms used to describe information supplied by the manufacturer
...
add Preview
Collegati
Allegati
|
Descrizione |
Lingua |
Dimensioni |
Downloads |
 |
|
EN |
860 kB |
0 |




































